Visit our Frequently Asked Questions Here.
The Cochrane Collaboration review of 17 randomised controlled trials involving 20,342 participants has contributed to recent debate about anti-amyloid therapies in Alzheimer’s disease. However, it is important to recognise that this dataset spans multiple generations of drug development and is not reflective of a single, uniform class of treatment.
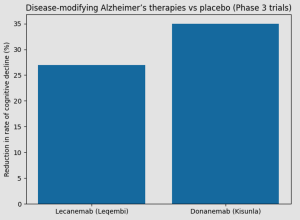
Of the studies included, only two trials relate to the modern, approved disease-modifying therapies now used in clinical practice – lecanemab (leqembi) and donanemab (kisunla) – together representing approximately 3,500 participants, or around 17% of the total evidence base. These pivotal Phase 3 trials demonstrated clinically meaningful outcomes, including significant slowing of cognitive and functional decline compared with placebo, and underpin current regulatory approvals.
The remaining majority of studies involve earlier investigational agents, many of which were discontinued after failing to demonstrate sufficient clinical efficacy despite targeting amyloid. Experts in the field have therefore raised concerns that pooling older first-generation compounds with newer, more targeted monoclonal antibodies risks obscuring treatment effects that are now clearly emerging in more advanced therapies.
As such, the interpretation of the overall findings must be considered in context. While earlier programmes were instrumental in advancing scientific understanding, they differ substantially in mechanism, efficacy and trial design from the next-generation therapies now available. The evidence base demonstrates that these newer treatments represent a meaningful step forward in slowing disease progression for patients in the earliest stages of Alzheimer’s disease.
Bart De Strooper, from the UK Dementia Research Institute at UCL, was particularly direct in his response, stating the review “does not clarify the evidence, it blurs it” and that “the flaw in this review is fundamental”. He emphasised that while many early programmes failed, newer antibodies have demonstrated modest yet real clinical benefit.
“the flaw in this review is fundamental”
For decades, a diagnosis of Alzheimer’s disease came with only symptomatic treatment options, nothing that targeted the disease pathology and an expectation of inevitable decline. Today, that narrative is changing. Advances in neuroscience have introduced a new generation of disease-modifying treatments that target the underlying biology of the disease, slowing progression in carefully selected patients and offering renewed hope for individuals and families affected by Alzheimer’s.
At Re:Cognition Health, we are proud to be at the forefront of this transformation, delivering these innovative treatments in clinical practice with lecanemab (leqembi) and donanemab (kisunla) and witnessing, on a daily basis, the meaningful impact they can have in preserving cognition, function and quality of life. By bridging cutting-edge research with specialist, real-world care, we are helping to redefine what is possible following an Alzheimer’s diagnosis.
THE EVIDENCE: Moving Beyond Symptom Management
Kisunla and Leqembi target amyloid plaques – abnormal protein build-ups in the brain widely recognised as a hallmark of Alzheimer’s disease. By actively reducing these plaques, they aim to slow the disease process itself, rather than simply easing its symptoms.
This represents a profound shift: from passive management to active intervention and the clinical data supporting these therapies is compelling.
In a large-scale Phase 3 trial of Donanemab (Kisunla) involving 1,736 participants demonstrated:
A 35% slowing of cognitive decline compared to placebo over 18 months
A 40% reduction in functional decline, preserving abilities such as managing finances, driving and maintaining hobbies
Among those in the earliest stages, a remarkable 60% reduction in disease progression
Crucially, Kisunla showed an 84% reduction in amyloid plaque levels, compared to just 1% in the placebo group. The treatment is delivered via a simple intravenous infusion every four weeks, providing a consistent and targeted approach.
Similarly, Lecanemab (Leqembi) has demonstrated meaningful benefits. In a trial of 1,795 participants with early-stage Alzheimer’s:
Patients receiving the drug experienced a 27% slower rate of cognitive decline over 18 months compared to placebo
While these figures may not represent a cure, they mark a significant clinical step forward – offering patients valuable additional time with preserved independence and quality of life.
Patient Perspective:
This progress is best understood not only through clinical data, but also through the lived experiences of patients who are among the first to access these new treatments. The following reflection offers a powerful insight into what this evolving landscape means in practice.
James Wood KC, patient at Re:Cognition Health comments:
“I am puzzled as to how the Cochrane review has reached such a firm conclusion when treatments such as donanemab are still in their early stages of use. As a 73-year-old practising barrister diagnosed with Alzheimer’s in October 2025, I was initially told I may be excluded from treatment with Kisunla due to my APOE E3/E4 status and a perceived increased risk of brain bleeds.
Kisunla is designed to remove amyloid plaques associated with Alzheimer’s disease, and it feels premature to dismiss its potential to delay disease progression meaningfully. These therapies are still very new, and their long-term impact on cognition is yet to be fully understood.
Given that donanemab is already in use internationally, I believe it is too early to restrict access in the UK. Wider availability, alongside careful monitoring, would allow more patients to benefit from this emerging class of treatment while generating valuable real-world data.
My view is simple: earlier testing and diagnosis are essential, so patients can access innovative treatments at the earliest and most effective stage of disease.”
Why Early Diagnosis Is Critical
One of the clearest messages emerging from this research is the importance of early diagnosis.
Both Kisunla and Leqembi are most effective when administered in the early stages of Alzheimer’s disease, before extensive damage has occurred. This means recognising symptoms early – such as subtle memory loss, changes in reasoning or difficulty with familiar tasks – and seeking specialist assessment without delay.
My view is simple: earlier testing and diagnosis are essential, so patients can access innovative treatments at the earliest and most effective stage of disease.
James Wood KC, A Patient at Re:Cognition Health
Early diagnosis not only opens the door to treatment but also allows individuals and families to plan, access support and take control of their care journey at a much earlier stage.
The Vital Role of Clinical Research
These breakthroughs have only been possible because of clinical research – and participation in clinical trials remains essential to advancing the field further. With every study undertaken, we learn more about the disease and helps refine effectiveness of treatments.
Re:Cognition Health plays a key role in global Alzheimer’s research, and is the leading centre internationally for recruiting participants onto studies – shaping the future of Alzheimer’s treatment. Clinical trials are free to join and offer participants access to cutting-edge therapies, similar to Kisunla and Leqembi, often years before they become widely available, alongside expert monitoring and support.
For many individuals, taking part in research is not only an opportunity to benefit personally, but also to contribute to progress that will help future generations.
The Benefits of Clinical Trials:
Access to early and highly accurate diagnosis using the most advanced investigative tools available
Potential to modify disease progression, with treatments aimed at slowing or halting decline
Early opportunity to access next-generation therapies that may not be widely available for several years
A chance to take a more active role in managing your health and future wellbeing
Ongoing, structured health monitoring throughout participation
Participation is free, with travel and related expenses often reimbursed
Contribution to important research that may benefit future generations
If you’re interested in learning more about the studies available for Alzheimer’s and other neurodegenerative conditions or interested in joining a study, contact our team
JOIN A TRIAL
Access to Treatment, Today
Encouragingly, these new-generation therapies are no longer confined to research settings.
Both Kisunla and Leqembi are now available through Re:Cognition Health centres in London, Bristol, Birmingham and Winchester. This means eligible patients can access some of the most advanced Alzheimer’s treatments currently available, delivered within a specialist, consultant-led environment.
A Turning Point in Alzheimer’s Treatment
The emergence of disease-modifying treatments marks a genuine turning point. While Alzheimer’s remains a complex and challenging condition, the outlook is no longer defined solely by inevitable decline.
Instead, we are entering an era where:
Progression can be slowed
Independence can be preserved for longer
Patients and families have meaningful options
The message is clear: doing nothing is no longer the only path.
At Re:Cognition Health, we are committed to ensuring patients benefit from these advances – through early diagnosis, access to innovative treatments and ongoing participation in world-class research.
If you are concerned about memory or cognitive changes, seeking advice early could make a significant difference.
 Visit our USA website
Visit our USA website